|
9/13/2023 0 Comments Sugar phosphate backbone ester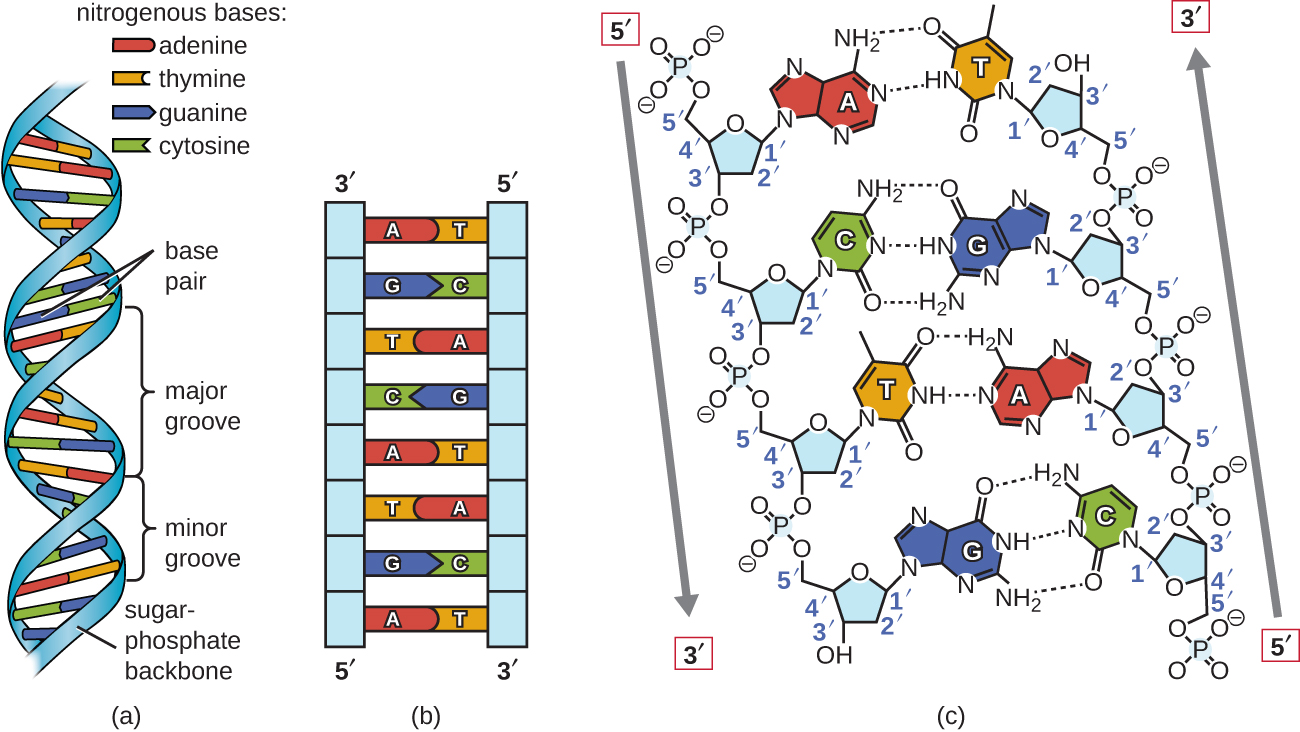
In contrast, DNA samples can be safely stored in aqueous buffer in a refrigerator, or in a freezer for longer-term storage. The problem of RNA decomposition is compounded by the fact that RNAase enzymes, which catalyze RNA hydrolysis, are present on the surface of human skin and are very stable, long-lived, and difficult to destroy. Take note of the 1' - 5' carbon numbering shown below for the ribose sugar - these numbers will be used frequently in the coming discussion. Researchers working with RNA have to be careful to store their samples at very cold temperatures, preferably freeze-dried or precipitated in ethanol, to avoid hydrolysis. Phosphate diesters play an absolutely critical role in nature - they are the molecular 'tape' that connect the individual nucleotides in DNA and RNA via a sugar-phosphate backbone. The 2' hydroxyl group is right next to the electrophilic phosphorus atom, poised in a good position to make a nucleophilic attack, breaking the RNA chain and forming a cyclic phosphate diester intermediate (see figure below). RNA nucleotides, unlike the deoxynucleotides of DNA, have a hydroxyl group at the neighboring 2' carbon. Why does hydrolysis occur so much more rapidly in RNA than in DNA? The answer has everything to do with the lowered entropic barrier to the reaction (you might want to quickly review the concept of entropy at this point). This does not present a physiological dilemma, because the function of RNA is to encode genetic information on a temporary rather than long-term basis. Unlike DNA, RNA is quite vulnerable to spontaneous hydrolysis in aqueous solution. 
Enzymes that hydrolyze the phosphate diester bonds in DNA are called nucleases, and we will learn more about them in section 9.8. As further phosphates are added, adenosine diphosphate and then adenosine triphosphate are formed.\)) in the active site. Addition of a phosphate to adenosine forms adenosine monophosphate. Thus, adenine attached to ribose forms the nucleoside adenosine. They are named by modifying the name of the nucleoside they contain together with the number of phosphates they contain. They can also play important roles in other cellular processes such as signalling between or within cells. These strong bonds form a sugar-phosphate backbone. They are used as the monomers in RNA and DNA, but are also components of important enzyme cofactors (see Ch 15), such as coenzyme A (CoA), flavin adenine dinucleotide (FAD), flavin mononucleotide (FMN), nicotinamide adenine dinucleotide (NAD), nicotinamide adenine dinucleotide phosphate (NADP) and adenosine triphosphate (ATP). and the phosphate of the next nucleotide. Nucleotides are made by adding a phosphate group to a nucleoside (Fig 9-2). Thus, the four nucleosides based on ribose are adenosine, guanosine, cytidine and uridine, and the four nucleosides based on deoxyribose are deoxyadenosine, deoxyguanosine, deoxycytidine and deoxythymine. What is the bond between pentose sugar and phosphate Nucleotides are joined together by covalent bonds between the phosphate group of one nucleotide and the third carbon atom of the pentose sugar in the next nucleotide. There are four types of nitrogenous bases in DNA. The building blocks of DNA are nucleotides, which are made up of three parts: a deoxyribose (5-carbon sugar), a phosphate group, and a nitrogenous base ( Figure 9.3 ). They are named by modifying the name of the base they contain. The phosphates and sugar molecules form the backbone of a DNA or RNA strand. Now let’s consider the structure of the two types of nucleic acids, deoxyribonucleic acid (DNA) and ribonucleic acid (RNA). Nucleosides are made by attaching a base to one of the sugars, either ribose or deoxyribose. The other family, the purines, have a structure based on two rings, and consist of adenine and guanine which are found in both DNA and RNA. In RNA the thymine is replaced by uracil. The pyrimidine bases have a single ring, with cytosine and thymine being found in DNA. The bases can be divided into two families based on the number of rings in their structure. FIGURE 9-1 The structures of the major bases found in nucleic acids.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
